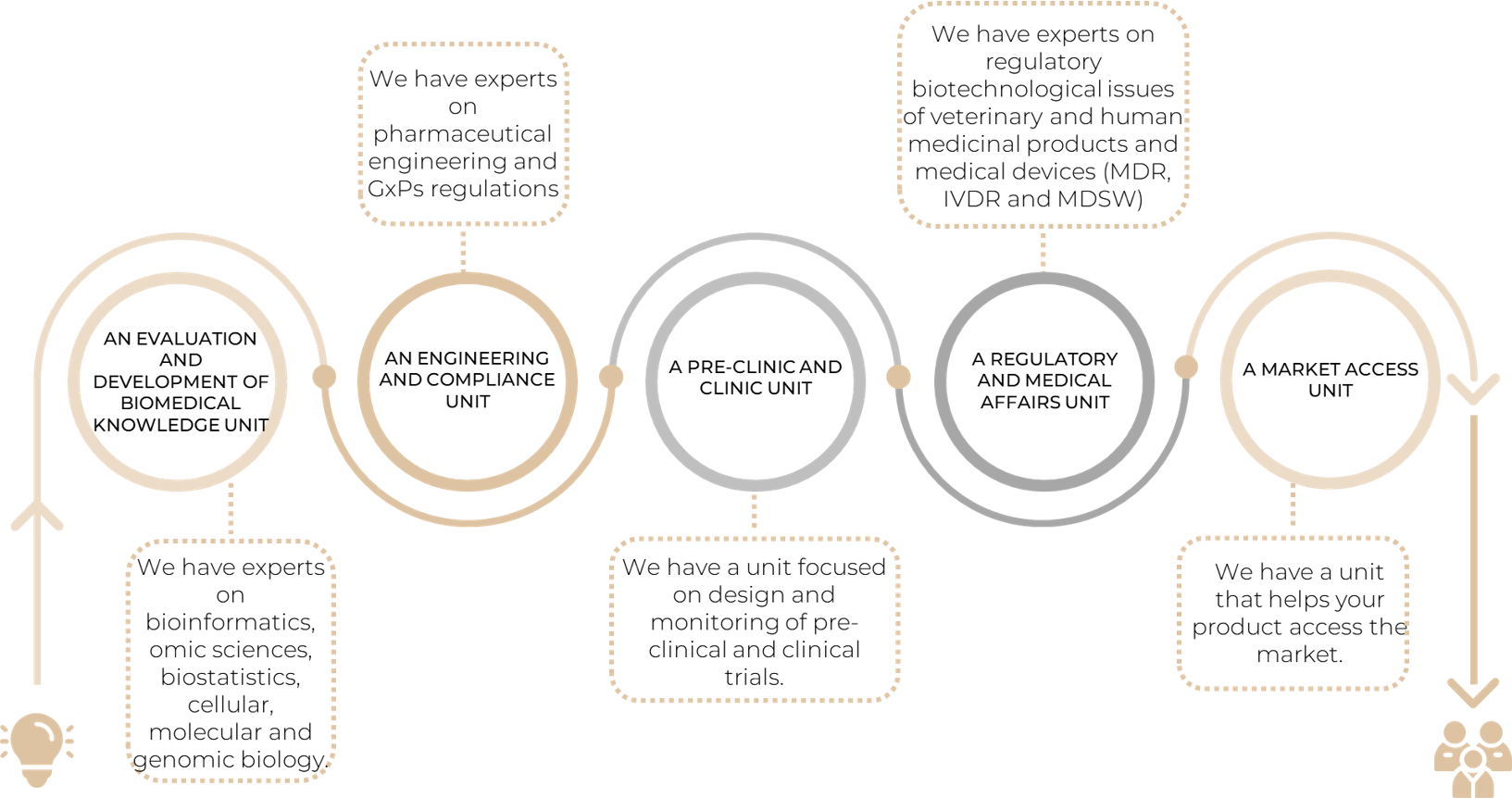
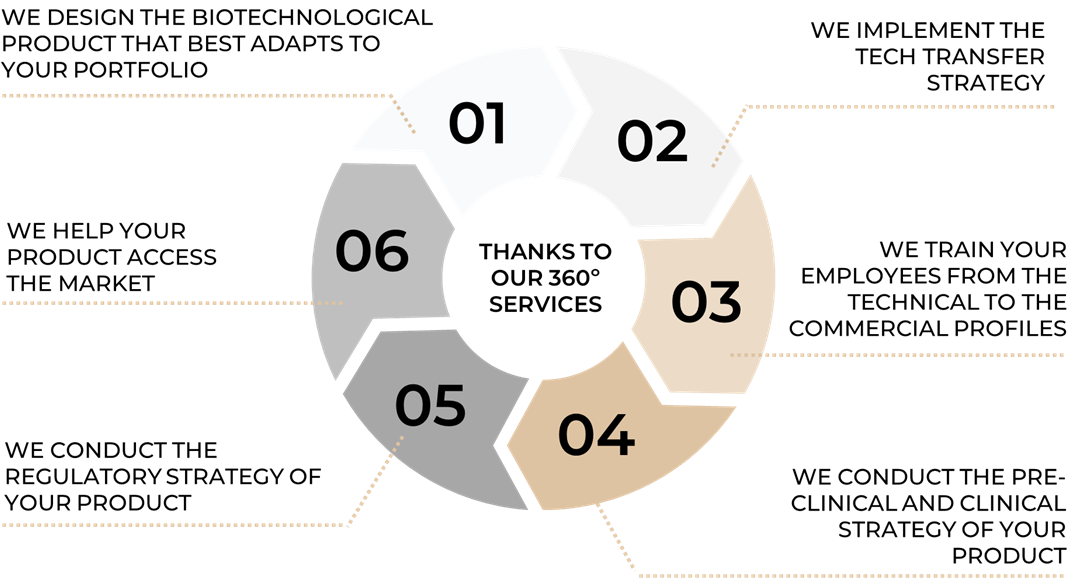
Thanks to the diversity of our team:
Systems of Standards for the Correct Manufacture of Medicines
Developed and implemented Standards Systems for Correct Manufacturing of Medications for human use in biopharmaceutical companies.
Auditorías de instalaciones y sistemas de calidad
Conducting audits of facilities and quality systems in production units in hospitals and healthcare clinics.
Audits of facilities and quality systems
Scientific advice on facilities and Good Manufacturing Practices
Offering scientific advice on facilities and Good Manufacturing Practices in companies specialized in the manufacture of Viral Vectors, plasmids and biomolecules.
GMP Training
We carry out GMP training applied to Advanced Therapies in various national and international hospitals.
GMP Training
Development of biotechnological products
Developing biotechnological products (AEMPS scientific advice, preclinical and clinical study, technical dossier, financial study) from biomolecules for both cosmetics and health products (MDR and IVDR).
Métodos analíticos y validaciones de proceso
Performing analytical methods and process validations for biopharmaceutical companies.
Analytical methods and process validations
Research projects
Advising and preparing research projects for CDTI.
Farmabiotec
We have launched Farmabiotec magazine.
Farmabiotec
Farmaforum
Organizing and coordinating Biotechforum at Farmaforum.
KONEXIO BIOTECH CONSULTING
CONNECT TO ADVANCE,
BE BIOTECH

Would you like to collaborate?
Let’s talk!